Amniotic–Chorionic Membrane Regenerative Therapy: A New Trend from Sports Medicine to Dental Soft Tissue Reconstruction
Amniotic–Chorionic Membrane Regenerative Therapy: A New Trend from Sports Medicine to Dental Soft Tissue Reconstruction
— Why the “amniotic membrane” technology favored by elite athletes is transforming the regenerative medicine landscape
📍 Latest News Spotlight
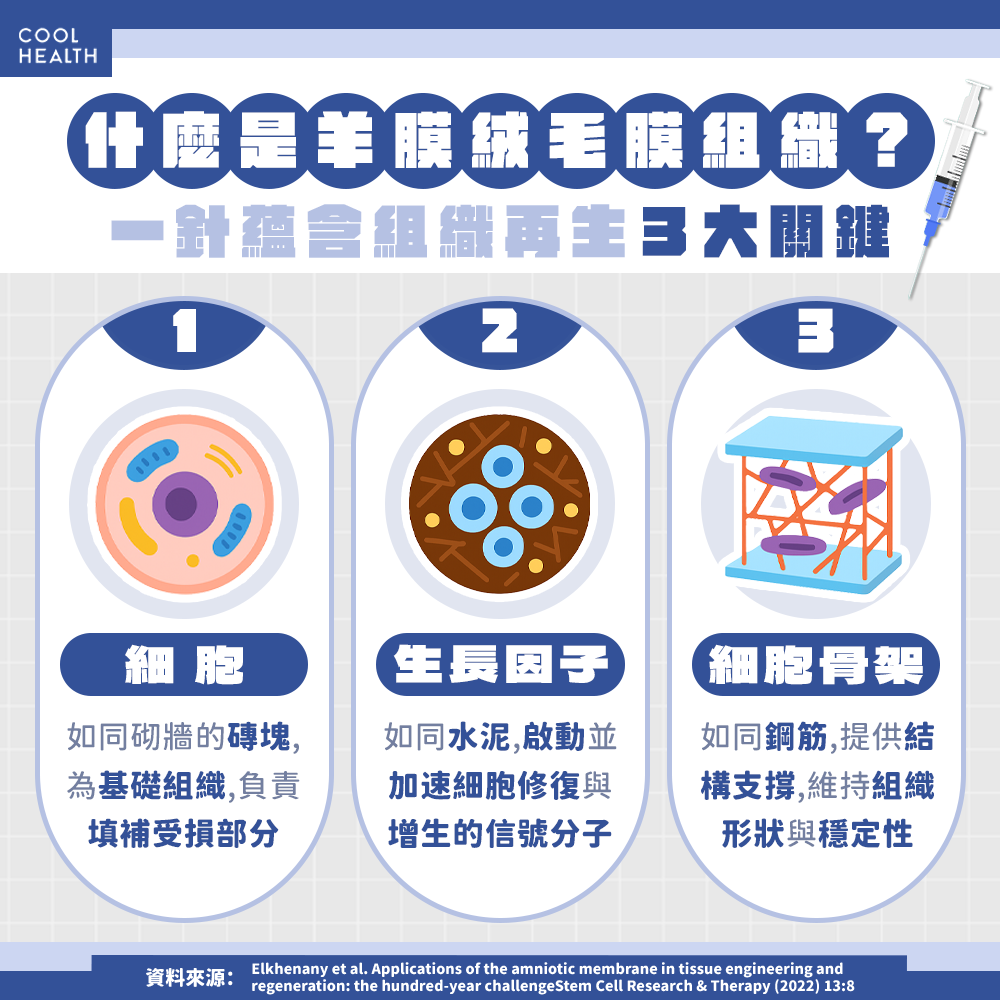
A recent FTV News report titled “Used by NBA and CPBL Players! Physicians Reveal Who Benefits from Amniotic–Chorionic Membrane Therapy” highlighted the growing medical use of amniotic–chorionic membranes derived from human placenta. These membranes contain abundant growth factors and extracellular matrix (ECM) components that promote tissue regeneration and modulate inflammation.
Clinicians noted that amniotic tissue can accelerate soft tissue healing, reduce scar formation, and prevent postoperative adhesions. As a non-cellular natural biomaterial with high safety and low immunogenicity, it is now recognized as one of the most advanced and reliable materials in regenerative medicine.
💡 A Turning Point in Regenerative Biomaterials: Extending from Sports Medicine to Dental Soft Tissue Repair
The advantages of amniotic membranes are not limited to orthopedics or sports medicine. Increasingly, dental clinicians have adopted these biomaterials for soft tissue regeneration, gingival coverage, and post-implant bone augmentation protection.
According to recent international reviews (e.g., Springer, 2025), amniotic membranes are proven to contain:
-
Multiple growth factors (EGF, FGF, TGF-β) that activate cellular repair responses
-
Natural collagen and ECM scaffolds that support cellular adhesion and regeneration
-
Anti-inflammatory and anti-adhesive properties that enhance postoperative stability and healing speed
These features give amniotic membranes distinct advantages in gingival thickening, root coverage, GBR membrane overlay, and composite use with PRP or bone grafts.
In short, this “from joint repair to dental regeneration” transition is opening new opportunities in the self-pay and advanced care markets.
🧩 Clinical Insights: Healing Efficiency Depends on More Than Bone Volume
Traditional bone grafting and soft tissue reconstruction have long focused on bone volume restoration. However, soft tissue integration and stability are the true determinants of long-term success.
Amniotic membranes contribute by:
-
Reducing postoperative inflammation and discomfort
-
Stabilizing the graft site while maintaining moisture and preventing adhesion
-
Guiding epithelial regeneration at an optimal rate
As a result, more clinicians now adopt a “Triple Regeneration Strategy” combining amniotic membrane + bone graft + PRP, achieving superior regenerative outcomes.
🚀 Future Outlook
Globally, amniotic-derived products are already established across four major clinical fields: sports medicine, dermatologic wound care, ophthalmology, and dentistry.
In Taiwan, since 2024, major medical centers—including Taipei Veterans General Hospital, Shin Kong Hospital, National Cheng Kung University Hospital, and Kaohsiung Medical University Hospital—have initiated clinical use of amniotic membrane products.
Moving forward, the next key step lies in bridging clinical adoption with patient education, bringing regenerative biomaterials from medical applications into the aesthetic and self-pay dental sectors, fostering a sustainable partnership between clinicians and biomedical innovators.
🔗 Further Reading
-
FTV News: “Used by NBA and CPBL Players! Physicians Reveal Who Benefits from Amniotic–Chorionic Membrane Therapy.”
-
Liberty Times Health: “Human Amniotic Tissue Injection—A Novel Therapy for Degenerative Arthritis.”
-
Springer Review: “The Human Amniotic Membrane and Its Applications in Regenerative Dentistry.”